Type IIa diamonds
Basic diamond types
Diamonds by presence of the natural impurities are divided into two main types, type I and type II.
Type I and Type II that are also divided into subtypes.
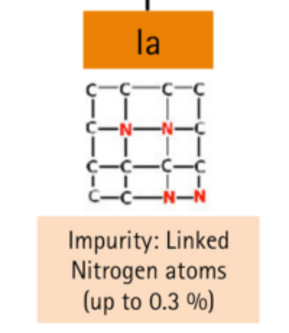
With aggregated (N) pairs
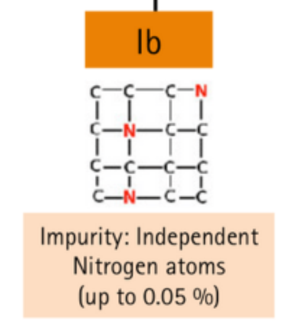
Isolated single (N) impurities
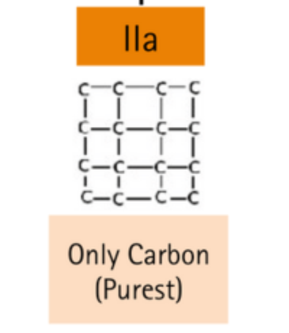
Without (N) or (B), impurities
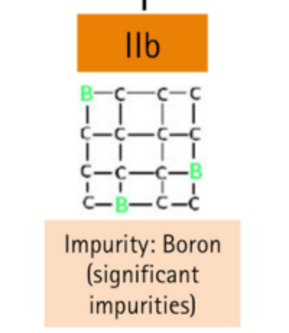
With (B) impurities
| Type I | Type II | ||
| Ia | Ib | IIa | IIb |




Basic diamond types table
| Type | Impurities | Color |
| Ia | Presence of nitrogen impurities, up to 0.3% | Colorless, pale yellow |
| Ib | Dispersed nitrogen atoms, up to 0.05% | Yellow, orange, brown |
| IIa | Pure carbon (almost, or devoid of impurities) | Colorless, yellow, brown, pink, red, magenta |
| IIb | Very low content of nitrogen impurities. Significant boron impurities. | Blue, gray |
We at LaBrilliante always have a wide selection of Type 2A diamonds on hand, ready to be shipped to you the next day.
Type I
Type I diamonds consist of carbon and nitrogen atoms.
If the atoms of nitrogen are in crystal pairs, and they do not affect the diamond's color, this type is called Type IaA.
If the nitrogen atoms are in larger quantities and impart a yellow to brown tint to a diamond, it’s Type IaB.
Type Ia diamonds: These diamonds contain isolated nitrogen atoms throughout the crystal structure. They can display a wide range of colors, from colorless to yellow or brown, depending on the concentration and arrangement of nitrogen atoms.
If the nitrogen atoms are not paired or grouped throughout the crystal in isolated sites, they give the stone an intense yellow or even brownish tint, this is Type Ib.

Type II
Type II diamonds don’t have or show any presence of nitrogen atoms in the crystal structure, and that leads them to colorless grade.
They’re divided into Type IIa and Type IIb.
Most of the lab-grown diamonds are Type IIa. They can also be found in gray, light yellow, light pink or light brown hues due to structural anomalies arising through deformation during crystal growth.
The price of a colorless diamond significantly depends on its type. The most rare and expensive diamonds in the world are all type IIa.
Not all D color diamonds will be type IIa.
According to the AGS Laboratory, over the past ten years, only 23% of diamonds graded D FL or D IF submitted to the laboratory have been recognized as type IIa diamonds.
Type IIb diamonds contain the boron atoms in the crystal structure, which makes most of the diamonds having a blue or grayish-blue color. They also have high electrical conductivity, as well as heat resistance and chemical stability.
You can find out the type of diamond from its grading certificate.
Wrap Up
Most diamonds are either colorless or have a yellowish tint, which means they contain nitrogen. These diamonds are type I and are further subdivided into type IaAB, IaA, IaB and Ib.
Colored diamonds contain almost no nitrogen and are classified as type II diamonds. Type II is also subdivided into IIa and IIb. Type IIa diamonds can be colorless, brown or pink, as they consist almost entirely of carbon and are the purest type of diamond.
Type IIb diamonds contain boron and are otherwise composed only of carbon; these diamonds are blue or gray in color.
Lab-grown diamonds can fall into various types, including Type IIa and IIb. However, not all lab-grown diamonds are categorized under these types, as they can also fall into other types, such as Type Ib or Type Ia, depending on the impurities present during their formation.